Viele Marken gehen davon aus, dass „medizinisch hochwertig“ automatisch sicher bedeutet. In Wirklichkeit, Der Begriff hat keine allgemeingültige rechtliche Definition. Die Sicherheit hängt davon ab, ob das Silikon frei von eingeschränkten Substanzen ist, richtig ausgehärtet, und durch Tests Dritter validiert.
In diesem Leitfaden wird erläutert, wie Silikone für empfindliche Haut auf chemischer und regulatorischer Ebene überprüft werden. Es beschreibt die REACH-Anforderungen, die Restrisiken D4/D5/D6, Auswahlkriterien für Klebstoffe, Dokumentationsstandards, und werkseigene Qualitätskontrollsysteme, die für vertretbare Sicherheitsansprüche erforderlich sind.
Was „medizinische Qualität“ bei der Silikonherstellung wirklich bedeutet

Der Begriff „medizinische Qualität“ wird in der Industrie häufig verwendet, ist jedoch keine eigenständige regulatorische Klassifizierung. In der Praxis, Damit sind in der Regel Silikonmaterialien gemeint, die so konzipiert sind, dass sie anerkannte Bioverträglichkeitsstandards wie ISO erfüllen 10993 oder USP-Klasse VI.
Für Konsumgüter, insbesondere Unterwäsche und Körperpflegezubehör, Die Sicherheit von medizinischem Silikon muss anhand chemischer Vorschriften wie dem REACH-Rahmenwerk der EU bewertet werden (EC 1907/2006 und Änderungen).
Ein Produkt kann nur dann als geeignetes Silikon für empfindliche Haut gelten, wenn es sowohl toxikologische als auch behördliche Vorgaben erfüllt.
Regulierungsrahmen für die Sicherheit von medizinischem Silikon
Die Reinheit des Materials allein definiert nicht die Sicherheit von Silikon in medizinischer Qualität; Durch die Einhaltung gesetzlicher Vorschriften wird die rechtliche und technische Grundlage für sichere Hautkontaktanwendungen geschaffen.
ERREICHEN
ERREICHEN (Anmeldung, Auswertung, Zulassung und Beschränkung von Chemikalien) ist die wichtigste Chemikalienverordnung der Europäischen Union. Es verlagert die Beweislast auf Hersteller und Importeure, Sie müssen nachweisen, dass die in Produkten verwendeten Stoffe sicher sind.
D4, D5, D6 Silikonbeschränkungen
Für Silikonmaterialien, Das kritischste Compliance-Problem betrifft zyklische Siloxane – allgemein als D4 D5 D6-Silikonverbindungen bezeichnet:
- D4 (Octamethylcyclotetrasiloxan)
- D5 (Decamethylcyclopentasiloxan)
- D6 (Dodecamethylcyclohexasiloxan)
Diese D4-D5-D6-Silikonverbindungen werden mit Umweltpersistenz und potenzieller Reproduktionstoxizität in Verbindung gebracht. Gemäß REACH Anhang XVII Beschränkungen, Ihre Konzentration ist in vielen Anwendungen begrenzt.
Warum minderwertiges Silikon das Reizungsrisiko erhöht
Minderwertiges Silikon ist nicht nur eine preisgünstige Wahl; Es handelt sich um ein chemisches Risiko. Im Gegensatz zu medizinischen Materialien, “billig” Silikon erreicht oft keine vollständige Polymerisation, Es bleiben reaktive Rückstände zurück, die die Sicherheit der Haut gefährden.
Unvollständige Polymerisation
Kostengünstiges Silikon entsteht oft durch verkürzte Aushärtungszyklen oder unzureichende Nachbearbeitung. Dies kann zurückbleiben:
- Restmonomere
- Nicht umgesetzte Siloxane
- Flüchtige Nebenprodukte
Diese niedermolekularen Verbindungen sind klein genug, um in die Hornschicht einzudringen, insbesondere unter Okklusion oder Hitze.
Für Produkte, die für längeres Tragen konzipiert sind, Okklusion verstärkt die Belichtung. Im Laufe der Zeit, Dies kann entzündliche Reaktionen auslösen, die üblicherweise als Kontaktdermatitis bezeichnet werden.
Fehlen von Migrationstests
Die Reinheit der Rohstoffe allein reicht nicht aus. Fertige Produkte müssen einem chemischen Migrationstest unterzogen werden, um sicherzustellen, dass eingeschränkte Substanzen bei normalem Gebrauch nicht auslaugen.
Für Silikon für empfindliche Haut, Migrationstests sollten simulieren:
- Längerer Hautkontakt
- Erhöhte Körpertemperatur
- Schweißexposition
Werden diese Tests nicht durchgeführt, entstehen Compliance- und Haftungsrisiken.
Erzielen Sie höhere Margen mit hochwertigen Brustwarzenabdeckungen

Die Rolle von Wacker-Kleber bei der Empfindlichkeitsprävention
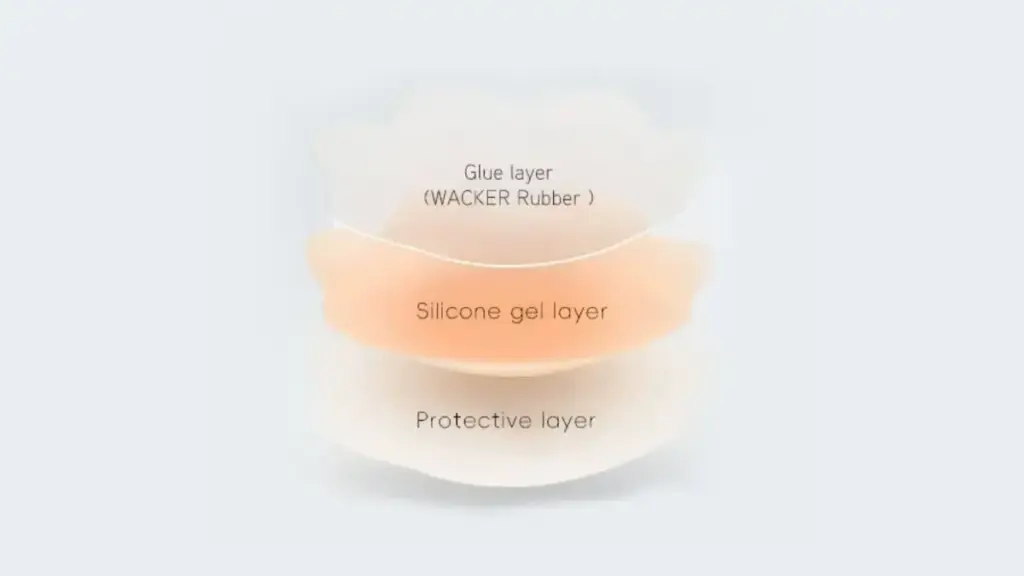
In tragbaren Silikonprodukten, Hautreaktionen hängen häufiger mit der Klebeschicht als mit dem Silikonkörper selbst zusammen. Ein instabiler Kleber kann Reste von Lösungsmitteln oder flüchtigen Verbindungen enthalten, die das Risiko von Reizungen bei längerem Tragen erhöhen.
Bei Xinke, Wir verwenden Silikonklebstoffsysteme der Wacker Chemie AG, da deren Rezepturen auf hohe Reinheit ausgelegt sind, Hautkontaktanwendungen bei sachgemäßer Verarbeitung. Diese Systeme sind darauf ausgelegt, Rest-D4 zu kontrollieren, D5, und D6-Inhalte und sind mit den REACH-Konformitätsanforderungen kompatibel.
Was bedeutet das praktisch??
- Geringeres Risiko flüchtiger Rückstände auf der Haut
- Stabile Verbindung bei Hitze und Schweiß
- Reduzierte Wahrscheinlichkeit von Rötungen oder Juckreiz während der Entfernung
Wir validieren die Klebstoffstabilität durch Migrationstests, um zu bestätigen, dass chemische Komponenten bei normalem Gebrauch innerhalb sicherer Grenzen bleiben.
Atmungsaktivität und Okklusionskontrolle
Okklusion ist ein Schlüsselfaktor für die Hautverträglichkeit. Wenn Silikonprodukte Wärme und Feuchtigkeit auf der Haut speichern, die Gefahr einer Mazeration, Rötung, und das Unbehagen nimmt zu – insbesondere bei längerem Tragen. Das Design aus echtem hypoallergenem Silikon muss sicheren Halt und Luftzirkulation in Einklang bringen.
Xinkes medizinische Materialnippelabdeckung ist klebstofffrei gefertigt, echte selbstklebende Struktur. Im Gegensatz zu lösungsmittelbasierten Klebeschichten, Dieses System beruht auf der inhärenten Klebrigkeit von gereinigtem Silikon und nicht auf dem Zusatz chemischer Klebstoffe. Dies verringert die Wahrscheinlichkeit einer klebstoffbedingten Empfindlichkeit und sorgt gleichzeitig für einen gleichbleibenden Halt.
Durch den Wegfall separater Leimschichten und die Optimierung der Oberflächenstruktur, Das Produkt behält die Haftung bei und ermöglicht gleichzeitig eine bessere Feuchtigkeitskontrolle. Für Marken, die Silikon für empfindliche Haut suchen, Dieses Gleichgewicht zwischen Atmungsaktivität und Stabilität trägt dazu bei, das Reizungsrisiko zu reduzieren, ohne die Leistung zu beeinträchtigen. Bleibt während der Bewegung sicher und behält gleichzeitig den natürlichen pH-Wert und Feuchtigkeitsgehalt der Haut bei.
Dokumentation: Was Marken anfordern müssen
Angaben wie „medizinisch geeignet“ oder „hypoallergen“ müssen mindestens durch technische Unterlagen belegt werden, Marken sollten anfordern:
SVHC-Screeningbericht
Bestätigt die Abwesenheit oder kontrollierte Konzentration eingeschränkter Stoffe gemäß REACH.
Testbericht zur chemischen Migration
Zeigt, dass Substanzen unter simulierten Verwendungsbedingungen nicht auslaugen.
Sicherheitsdatenblatt (Sicherheitsdatenblatt)
Bietet Offenlegung der chemischen Zusammensetzung und Gefahrenklassifizierung.
Ein Sicherheitsdatenblatt ist keine Zertifizierung. Es handelt sich um ein vom Lieferanten ausgestelltes Offenlegungsdokument. Es muss durch unabhängige Laborberichte gestützt werden.
Tests durch Dritte (Z.B., SGS)
Unabhängige Inspektionsstellen wie SGS bieten eine Laborvalidierung an:
- Schwermetallgehalt
- SVHC-Konformität
- Leistung der chemischen Migration
Für den EU-Markteintritt, Die Dokumentation muss mit der Europäischen Chemikalienagentur übereinstimmen (ECHA) Anforderungen.
Ohne diese technische Datei, Sicherheitsansprüche lassen sich bei Audits oder behördlichen Überprüfungen nur schwer verteidigen.
Qualitätskontrolle im Werk: Sicherstellen, dass flüssiges Silikon keinerlei Verunreinigungen enthält

Um die Sicherheit von medizinischem Silikon zu erreichen, ist ein strukturiertes QC-System erforderlich.
Rohstoffverifizierung
Jede eingehende Silikoncharge sollte durchgeprüft werden:
- Überprüfung des Lieferanten-Sicherheitsdatenblatts
- ECHA-Registrierungsbestätigung
- Rückverfolgbarkeit auf Chargenebene
Materialien ohne vollständige Dokumentation sollten nicht in die Produktion gelangen.
Analytische Tests
Fortgeschrittene Hersteller implementieren:
- GC-MS zur Detektion flüchtiger Siloxane
- ICP-MS für das Schwermetall-Screening
- Validierungstests nach der Aushärtung
Diese Methoden erkennen restliche D4/D5/D6-Silikonverbindungen und stellen sicher, dass die Konzentrationswerte innerhalb der gesetzlichen Grenzwerte bleiben.
Rückverfolgbarkeit und Aufbewahrung von Chargen
Eine konforme Einrichtung unterhält:
- Aufzeichnungen zur Chargenrückverfolgbarkeit
- Rückstellmuster
- Revisionsfähige Dokumentation
Dies schützt sowohl den Hersteller als auch den Markeninhaber im Falle behördlicher Untersuchungen oder Streitigkeiten über Produktansprüche.
Abschluss
Die Sicherheit von medizinischem Silikon hängt von der dokumentierten REACH-Konformität ab, kontrollierte D4/D5/D6-Pegel, stabile Klebesysteme, und verifizierte Migrationstests. Etiketten allein reichen nicht aus – Beweise sind erforderlich.
Bei Xinke, Wir kombinieren Hochreinheit, Nicht klebendes, selbstklebendes Silikon mit validierten Tests und vollständiger Konformitätsdokumentation zur Unterstützung zuverlässiger Silikone für empfindliche Haut. Wenn Sie transparente technische Dateien oder Produktmuster benötigen, Kontaktieren Sie unser Team um Zertifizierungen zu überprüfen und Ihre zu besprechen OEM/ODM-Projekt.
Häufig gestellte Fragen
Ist medizinisches Silikon automatisch sicher für empfindliche Haut??
NEIN. Sicherheit hängt von der Einhaltung gesetzlicher Vorschriften ab, chemische Reinheit, und Migrationstests – nicht nur auf dem Etikett.
Können Brustwarzenabdeckungen einen Ausschlag verursachen??
Ja. Es kann zu Reizungen kommen, wenn Silikon oder Klebstoff Rückstände von Chemikalien wie z. B. eingeschränktes D4 enthalten, D5, oder D6-Verbindungen. Produkte ohne REACH-Konformität und ohne Migrationstests bergen ein höheres Risiko für Hautreaktionen.
Was ist der Unterschied zwischen lebensmittelechtem und medizinischem Silikon??
Silikon in Lebensmittelqualität entspricht den Lebensmittelkontaktvorschriften wie der EG 1935/2004. Medizinisches Silikon ist in der Regel auf Biokompatibilität ausgelegt. Beide müssen beim Verkauf in der EU die REACH-Chemikalienbeschränkungen erfüllen.
Enthalten Brustwarzenabdeckungen Latex??
Die meisten Nippelabdeckungen aus Silikon sind latexfrei. Jedoch, Die Materialzusammensetzung hängt vom Hersteller ab. Überprüfen Sie vor der Beschaffung immer das Produktspezifikationsblatt und die Sicherheitsdokumentation.
So erkennen Sie, ob ein Hersteller giftigen Kleber verwendet?
Fordern Sie Dokumente zur REACH-Konformität an, Ergebnisse des SVHC-Screenings, und Migrationstestberichte. Transparente Lieferanten stellen vollständige technische Unterlagen zu Silikon- und Klebstoffkomponenten zur Verfügung.
Auf welche Zertifizierungen sollte ich achten??
Überprüfen Sie zunächst die REACH-Konformität. Zusätzliche Dokumentation kann eine CE-Kennzeichnung umfassen, RoHS-Konformität, und Migrationstestberichte zur Validierung der Hautkontaktsicherheit.
